Discuss the below:
Heat flow and work done by an ideal gas
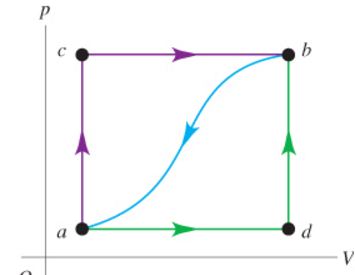
Q: When a system is taken from state a to state b in the figure View Figure along the path acb, the amount of heat that flow into the system is 95.0 J and the work done by the system is 57.0 J.
A) How much heat flows into the system along path adb if the work done by the system is 10.0 J?
B) When the system is returned from b to a along the curved path, the absolute value of the work done by the system is 32.0 J. How much heat does the system liberate?
C) If the internal energy is zero in state a and 13.0 J in state d, find the heat absorbed in the processes ad.
D) Find the heat absorbed in the processes db.